
Comment calculer un pourcentage massique 13 étapes
4: Atomic Structure 4.5: Mass Ratio Calculation

In the chemical analysis of a rock the mass ratio of two radioactive isotopes is found to YouTube
The mass-to-charge ratio ( m / Q) is a physical quantity relating the mass (quantity of matter) and the electric charge of a given particle, expressed in units of kilograms per coulomb (kg/C). It is most widely used in the electrodynamics of charged particles, e.g. in electron optics and ion optics .
CHEMISTRY 11 ISOTOPES AND ATOMS October 26, 2010
What is mass ratio in chemistry? Stoichiometry: Stoichiometry refers to the calculations made to find amount of reactants and products in a chemical reaction. Stoichiometry is based on the law of conservation of mass, where the total mass of reactants will have to produce an equal amount of products, supposing the reaction has a 100% reaction.

Mass Ratios YouTube
Step 1: List the known quantities and plan the problem. Known Compound A = 4.08 g Cu and 2.28 g Cl Compound B = 7.53 g Cu and 8.40 g Cl Apply the law of multiple proportions to the two compounds. For each compound, find the grams of copper that combine with 1.00 g of chlorine by dividing the mass of copper by the mass of chlorine.

Charge to mass ratio of electron proof Charge to mass ratio mathematical proof F Sc Chemistry
Mass Ratios, Percent Composition & Empirical Formulas Quiz. This online quiz is intended to give you extra practice in determining mass ratios, percent compositions and empirical formulas of a variety of chemical compounds. Select your preferences below and click 'Start' to give it a try! Number of problems: 1. 5.

uchopenie ideológie Cena how to calculate mass percent odvolanie vitajte inzerovať
In chemistry, mass ratio, often called "percent composition by mass," is the proportion of a particular molecule that consists of each that molecule's constituent elements.

Quantum Gravity and the Holographic Mass Ratio Description The New Energy Industry
A mass spectrometer ionizes atoms and molecules with a high-energy electron beam and then deflects the ions through a magnetic field based on their mass-to-charge ratios ( m / z ). The mass spectrum of a sample shows the relative abundances of the ions on the y-axis and their m / z ratios on the x-axis. If z = 1

Mole Ratio Definition and Examples
Mass Percent Equations and Indicator Words. The mass percent of a solution is defined as the ratio of the mass of solute that is present in a solution, relative to the mass of the solution, as a whole. Additionally, because this type of concentration, which is typically calculated for solid- and liquid-phase solutions, is expressed as a percentage, this proportion must be multiplied by 100, as.

Mass charge ratio Science, Chemistry, Atoms, Atoms Ions ShowMe
Mass Ratio Here the ratio of the masses of molecules or atoms can be calculated. Please enter the empirical formula of two molecules, the entry format is similar to the Round to Molecular mass X: u Molecular mass Y: u Examples: The water molecule H 2 O has a mass ratio of oxygen to hydrogen of 7.94.

Structure of Atom Class 11 Chemistry Charge to Mass Ratio of Electrons YouTube
No headers. In mass spectroscopy, the mass-to-charge ratio (symbols: m/z, m/e) of a cation is equal to the mass of the cation divided by its charge. Since the charge of cation formed in the mass spectrometer is almost always +1, the mass-to-charge ratio of a cation is usually equal to the mass of the cation.

Unit 4 Using mass ratios to predict formulas YouTube
The mass ratio of copper per gram of chlorine in the two compounds is 2:1. For a given mass of chlorine, compound A contains twice the mass of copper as does compound B. For each compound, find the grams of copper that combine with 1.00 g of chlorine.

Charge to Mass Ratio Structure of the Atom CBSE Class 11 Chemistry YouTube
Where: Mass Ratio (MR) is the ratio of the mass of Substance A to the mass of Substance B, typically expressed as a decimal or fraction. Mass of Substance A (MA) is the mass of the first substance in the mixture or reaction, typically measured in grams (g), kilograms (kg), or other appropriate units.

9th Class Chemistry Chapter 6 Volume over Mass Ratio YouTube
In chemistry, the mass fraction of a substance within a mixture is the ratio (alternatively denoted ) of the mass of that substance to the total mass of the mixture. [1] Expressed as a formula, the mass fraction is: Because the individual masses of the ingredients of a mixture sum to , their mass fractions sum to unity:

PPT Chemistry Review PowerPoint Presentation, free download ID5678406
We can balance the equation by placing a 2 in front of NaOH (so that there are 2 Na atoms on each side) and another 2 in front of H A 2 O (so that there are 6 O atoms and 4 H atoms on each side). Doing so gives the following balanced equation: 2 NaOH ( a q) + H A 2 SO A 4 ( a q) → 2 H A 2 O ( l) + Na A 2 SO A 4 ( a q)

chargemass ratio of electron, standard 11 chemistry chapter2 Gujarati medium Part2 YouTube
Calculating mole ratios Remember, stoichiometry is the study of mass relations. To master it, you need to be comfortable with unit conversions and balancing equations. From there, the focus is on mole relationships between reactants and products in a chemical reaction. Mass-Mass Stoichiometry Problem
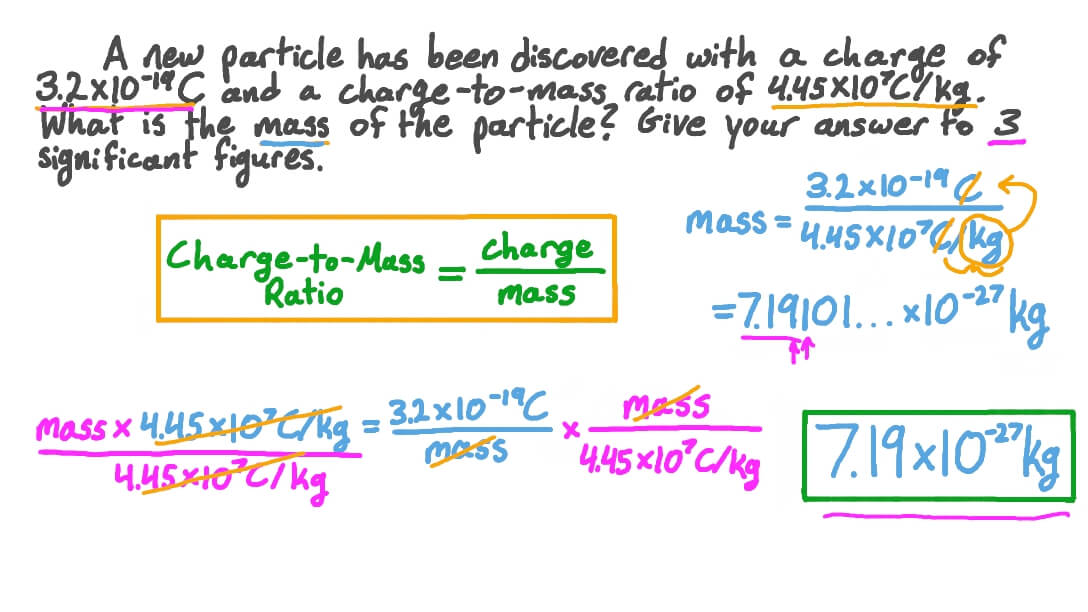
Question Video Using the ChargetoMass Ratio to Find the Mass of a Particle Nagwa
A teaching video on Mass Ratio used in the 'Global Climate Change' module at The University of Texas at Austin.